by Catherine M. Haug, January 2008 (some content moved from Saturated Fats: The Good Rap, originally from Foods About on iWeb site)
(Photo, right, from Wikimedia commons (8a)
Dietary fats are a popular topic these days, especially with the resurgence of the high-fat, low-carb diet in popular culture. The word “fat” makes us look down at our bellies; we think about butter, cooking oils, shortening and other dietary fats. But just what is a fat, beyond a high-calorie food? This article is an into to the chemistry of fats.
- Includes: 1. Intro to Fats and Fatty Acids; 2. Degree of saturation; 3. cis and trans configurations; 4. Omega designations; 5. Glycerides or fats
- See also: 1. Diet & Health Menu; 2. About Fats, an Introduction.
Are fats fattening?
We blame our tummy-fat on too much dietary fat, that is just not true. Dietary fat is a result of too much sugar in our blood – sugar from sugary and starchy foods – which typically results from our body’s cells being unable to take-up sugar, even when they are hungry. So the liver converts the excess sugar into storage fat, some of which remains in the liver, but the bulk it is sent to our fat cells (adipose tissue) to be stored.
The inability of cells to take-up sugar is a result of a problem known as insulin resistance (insulin is the key that ushers sugars into our cells). But that is another topic. (Refer to my Insulin Resistance articles (in iWeb Health-Disease section)
It’s also important to note that our bodies can make certain fats, primarily in the liver.
Fats and Fatty Acids
A fatty acid, the simplest of fats, is comprised of a hydro-carbon chain, with a carboxyl group (acidic COOH) at one end. The hydrocarbon chain is hydrophobic, meaning that it will not mix with water; the acid group is hydrophylic, meaning it is attracted to, and forms weak bonds with, water. However, the fatty acid as a whole is insoluble in water and is thus part of the family of lipids (substances insoluble in water).
This is an example of a fatty acid: the 18-carbon, saturated, stearic Acid which has 18 carbons in the chain. The point of each angle in the chain is a carbon with 2 hydrogens attached, except the left-end carbon has 3 hydrogens, and the right-end carbon has no hydrogens; instead, it is a COOH group (double-bonded to an oxygen and single bonded to an alcohol or OH group)(image from Wikimedia commons (8)):
Fatty acids can be categorized in several ways: by
- Degree of saturation: saturated (no double bonds between carbons), mono-unsaturated (only one carbon-to-carbon bond is a double bond), and polyunsaturated (more than one carbon-to-carbon bond is a double bond);
- Origin (animal vs vegetable); and
- Length of the hydrocarbon chain (short, medium and long-chain).
In nature, fatty acids are usually bound to something else to form:
- Glycerides: Up to 3 fatty acids may be linked together via a glyceride backbone, forming a type of fat known as glycerides or acylglycerols (refer to About fats, an introduction for more).
- Cholesteryl esters: A fatty acid conected to a cholesterol molecule via an ester bond
- Phospholipids: A fatty acid conected to a phosphate backbone
- Glycolipids: A fatty acid connected to a sugar molecule
(Refer to the Lipids articles (in iWeb Health-Metabolism section) for pictures and more).
Degree of Saturation
See Wikipedia article: Fatty Acids2 (8A) for great diagrams and more. Note that a single line (—) represents a single bond; and a double line (=) represents a double bond in the diagrams.
Saturated: Fatty acid chains that contain only single bonds between carbon atoms (C-C), and all 4 carbon-atom linkages are filled with single bonds to hydrogen atoms, or other carbon atoms in the chain. These are very stable, and seldom go rancid (become oxidized). They are also solid or semisolid at room temperature. Our bodies can get them from the diet, or manufacture them in the liver from carbohydrates. (Lauric acid is an exception because it can only be made in mammary glands, and thus is considered an essential fat that must be obtained from the diet). See diagram, above, of stearic acid for an example of a saturated fatty acid.
Monounsaturated: Fatty acid chains that contain one double bond between carbon atoms (C=C); all the other carbon-to-carbon bonds in the chain are single. A double bond involves two pairs of electrons, leaving only 2 other linkage points on each carbon to connect with hydrogen atoms, or with other carbon atoms in the chain. These natural fats have a kink in the chain at the point of the double bond, and thus do not pack together as tightly as saturated chains, making them liquid at room temperature. Monounsaturated fats are relatively stable, and do not go rancid easily. Our bodies can get them from the diet, or manufacture them from saturated fats.
Polyunsaturated: fatty acid chains contain two or more double bonds between carbon atoms (C=C).
Here are two examples of essential poly-unsaturated fatty acid molecules: linoleic acid (has 2 carbon-to-carbon double bonds) and linolenic acid (has 3 carbon-to-carbon double bonds) are examples (diagrams originally from reference 3, but the link no longer works). Note that all the double bonds in these examples are”cis.” See below for an explanation of Omega-3 and Omega-6 fatty acids.
These natural fats (cis) have a kink at each double bond and do not pack well at all. Their chemically altered (trans) counterparts do not have that kink, and so pack together as tightly as saturated fats, and are thus solid at room temperature (see double bond configurations, below, for more). Because of this, natural polyunsaturated fats are liquid even when refrigerated. They are also highly reactive (easily oxidized); the more double bonds in the chain, the more reactive the fat. While our bodies can manufacture most polyunsaturates from other fats, two must be obtained from the diet: linoleic and linolenic acids, and are thus termed essential fats.
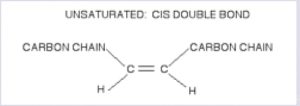
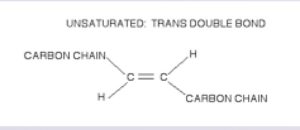
Cis and Trans configurations
The double-bonds in unsaturated fats have two possible configurations: cis and trans, as illustrated below (sketches by Cat).
- The cis configuration is considered “natural” because plants and animal can make them. In nature, polyunsaturated fatty acids are usually found in the cis or bio-active form, which means that both hydrogen atoms at the double bond are on the same side, thus exposing the double bond as a site where enzymes can work.
-
The trans configuration is considered “unnatural” or “chemically-altered” as a result of the hydrogenation process; for example, the trans fats in margarine. When polyunsaturated fats are hydrogenated, the hydrogen atoms at the double bond are on opposite sides. This is called the trans form, and is not bio-active (with only a few exceptions; see below). This bond does not cause a kink but rather a rigidity at the bond site, making it difficult for enzymes to work.
I do not intend to discredit the importance of polyunsaturated fats in food. I do intend to discredit hydrogenated and partially hydrogenated polyunsaturated fats in foods.
There are a few natural trans fatty acids, but by far the majority of trans fatty acids are man-made and cause great harm. One natural trans fatty acid is vaccenic acid, found in the cream fraction of dairy milk, and also in cheese and yogurt; another is palmitoleic acid in sea buckthorn and macadamia nuts.
Omega designations:
The number following ‘omega’ indicates the position of the last double-bond in the hydrocarbon chain (start counting the carbon-to-carbon bonds at the end opposite the acidic COOH end, to find the #3, #6, #7 and #9 position as described below). That position has a lot to do with the functionality of the molecule. (7) In the following descriptions, the “last double bond) is furthest from the carboxyl (COOH) group.
See the linoleic and linolenic diagrams above for examples.
- Omega-3 fatty acids are usually poly-unsaturated, with the last double bond in the 3rd position; linolenic acid, above is an example. With few exceptions, Omega-3s are anti-inflammatory. The most important are alpha-linolenic acid (ALA), eicosapentaenoic acid (EPA), and docosahexaenoic acid (DHA). they are key to neurological development, Animal sources include many fish such as salmon, herring, halibut and sardines, and also cod liver oil; plant sources include flax and hemp seeds, walnuts, pecans and hazelnuts. (5)
- Omega-6 fatty acids are usually poly-unsaturated, with the last double bond in the 6th position; linoleic acid, above, is an example. With few exceptions, Omega-6s are pro-inflammatory. Common omega-6s are linoleic acid (LA) and arachidonic acid (AA). Common food sources are safflower, corn, cottonseed and sunflower oils. (5)
- Omega-7 fatty acids have the last double bond in the 7th position, and that bond is trans. Sea buckthorn berry is a common source (but not sea buckthorn seed oil). Macadamia nuts are rich in palmitoleic acid, another omega-7. (6)
- Omega-9 fatty acids are mono-unstaturated fats with the single double bond in the 9th position. Oleic acid is the most common Omega-9. They are common in olive oil, and in animal fats such as lard .(5)
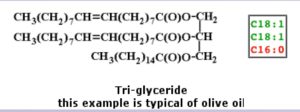
Glycerides or Fats
“Fats” generally means tri-glycerides, as this is the form found in fatty tissue. It is formed of a glycerol backbone (3-carbons) with a fatty acid attached to each of those 3 carbons (image, below from reference 4):
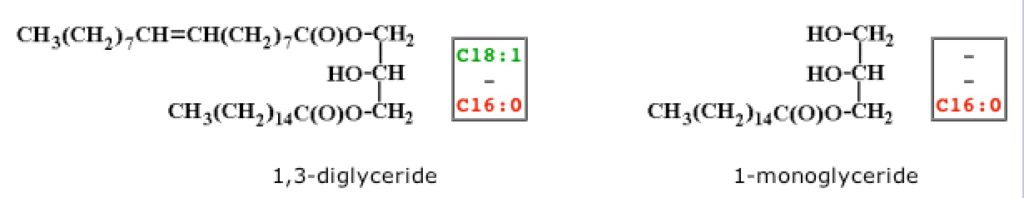
However, mono and di-glycerides are also present during fat metabolism. These have only one or two of the glycerol carbons linked to a fatty acid. In nature, mono-glycerides have the fatty acid linked to an end carbon; di-glycerides have the fatty acids linked to both end carbons, leaving the center carbon unlinked. However, many chemically manufactured mono and di-glycerides used in foods have a fatty acid on the center carbon, and this configuration causes problems for biological systems, such as the human body. Here are examples of mono and diglycerides (image, below from reference 4):
References
- Molecular Biology II: Lipoproteins from Renssalaer Polytechnic Institute
- chemistry.about.com/library/graphics/blchol.htm
- womega3sealoil.com/Chapter2a.html
- wscientificpsychic.com/fitness/fattyacids1.html
- hbuiltlean.com/2013/11/27/omega-3-6-9/
- fattyacidshub.com/fatty-acids/omega-7
- hdrsarahbrewer.com/food/omega-3-omega-6-omega-7-omega-9-more
- Wikipedia and wikimedia commons: